A brief explain why the Nd and Sr isotope composition of the Earth's continental crust and upper depleted mantle are so different
© J.A. Pfänder, Freiberg
To understand this, we need to know that the element Sm consists of several stable and unstable isotopes, and among the unstable ones, the 147Sm decays to stable 143Nd by an α-decay. This decay is shown in the diagram as blue curve. Note the extremely slow decay rate, which is due to the extremely small decay constant (i.e., very large half life).
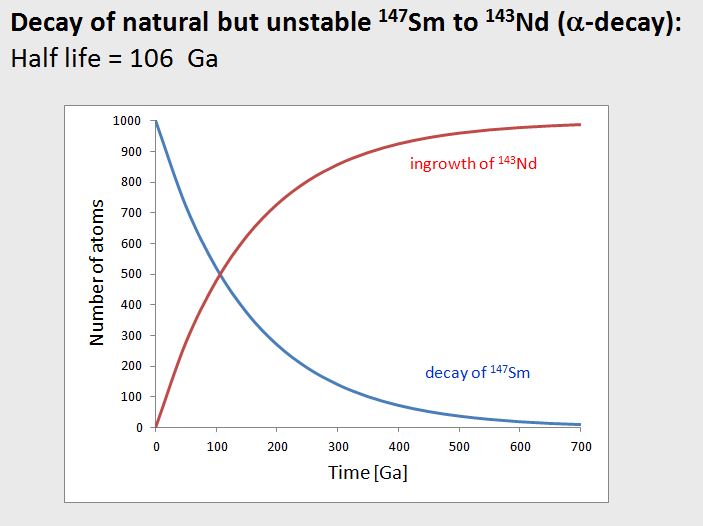
Now consider that any atom of 147Sm that decays ends up becoming a stable 143Nd isotope. So, as the abundance of 147Sm in rock or mineral decreases, the abundance of 143Nd in this rock or mineral increases at the same rate. This is the red curve in the diagram.
If we then consider the 143Nd/144Nd isotope ratio in a rock or mineral, this ratio will increase as a function of time due to the radiogenic ingrowth of 143Nd. In this context it is very important to know that 144Nd is a stable isotope, so its concentration in a rock or mineral will remain constant over time. Now, the important point here is that the higher the Sm/Nd ratio of a rock or mineral, the stronger is the increase in 143Nd/144Nd in this rock or mineral over time.
As the Nd istope composition of the present-day continental crust is unradiogenic, that means, the 143Nd/144Nd is low, the continental crust needs to have had a low Sm/Nd ratio since a longer time. As the Earth's present-day depleted upper mantle (as sampled by mid-ocean ridge basalts) is radiogenic in Nd, this implies the opposite, namely that the Earth's upper mantle needs to have had a high Sm/Nd ratio since a longer time.
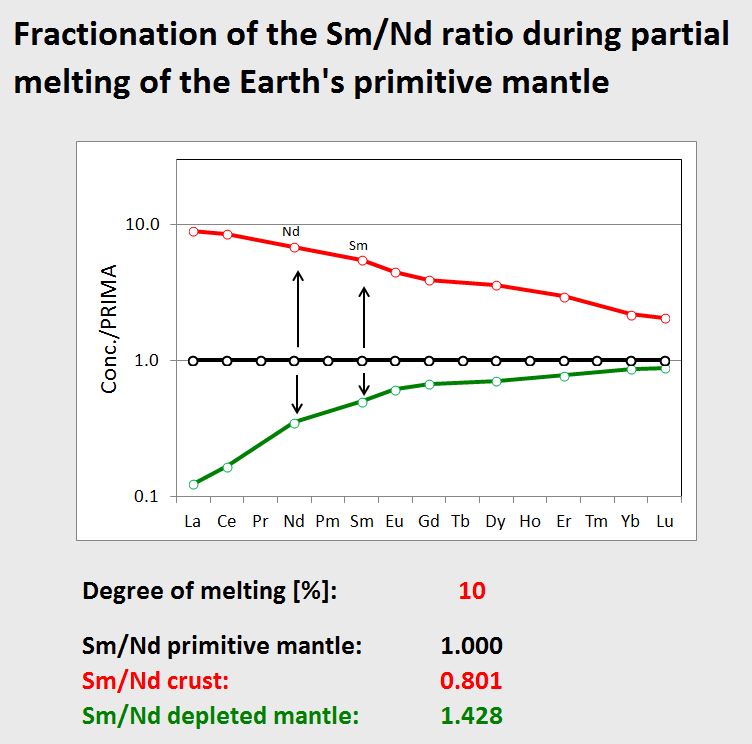
So, lets see what the reasons are that separate (or fractionate) Sm and Nd during crust formation.
Let the black line in the left diagram be the REE composition of the primitive mantle, that is the composition of the silicate portion of the Earth after its formation about 4.5 Ga ago. As we use compositions here that are normalised to the primitive mantle, the concentration for each REE is simply 1.
If we now generate a partial melt from this primitive mantle, and if we assume 10% of partial melting, we will end up with a magma with a REE pattern that is shown as red curve in the diagram. As you can see, Nd is enriched in this melt over Sm, and the normalised Sm/Nd ratio in our example will be 0.801, i.e. low. If we assume that the continental crust was formed by such a very simplified process, then this explains why the continental crust has developed an unradiogenic Nd-isotope compsiton over time.
What about the residue, i.e. the depleted mantle that lost this melt? Its composition is shown as green curve in the second diagram.This residue is depleted in light-REE, and as Nd is more incompatible than Sm during mantle melting, the Nd concentration is lower than the Sm concentration. This means that the residue has a high Sm/Nd ratio of 1.428 in this case, and thus the depleted mantle will develop to a radiogenic Nd-isotope compositon over time.
In summary, partial melting over the Earth's history is the reasons why the Earth's continental crust and depleted mantle have different Nd-isotope compositions, and exactly the same principles are valid for all other radioactive decay schemes (Lu-Hf, Rb-Sr, U-Th-Pb, Re-Os, ...).

